Blogs

Is Your QMS Ready for an Audit? A Practical 2026 Health Check for APAC Pharma & Biotech
May 28, 2026
If youre reading this with an inspection notice already on your desk, heres the uncomfortable truth: by the time the regulator arrives, your QMS is whatever it has been for the past two years. You cant paper over that gap in three weeks. But if yo...
Learn more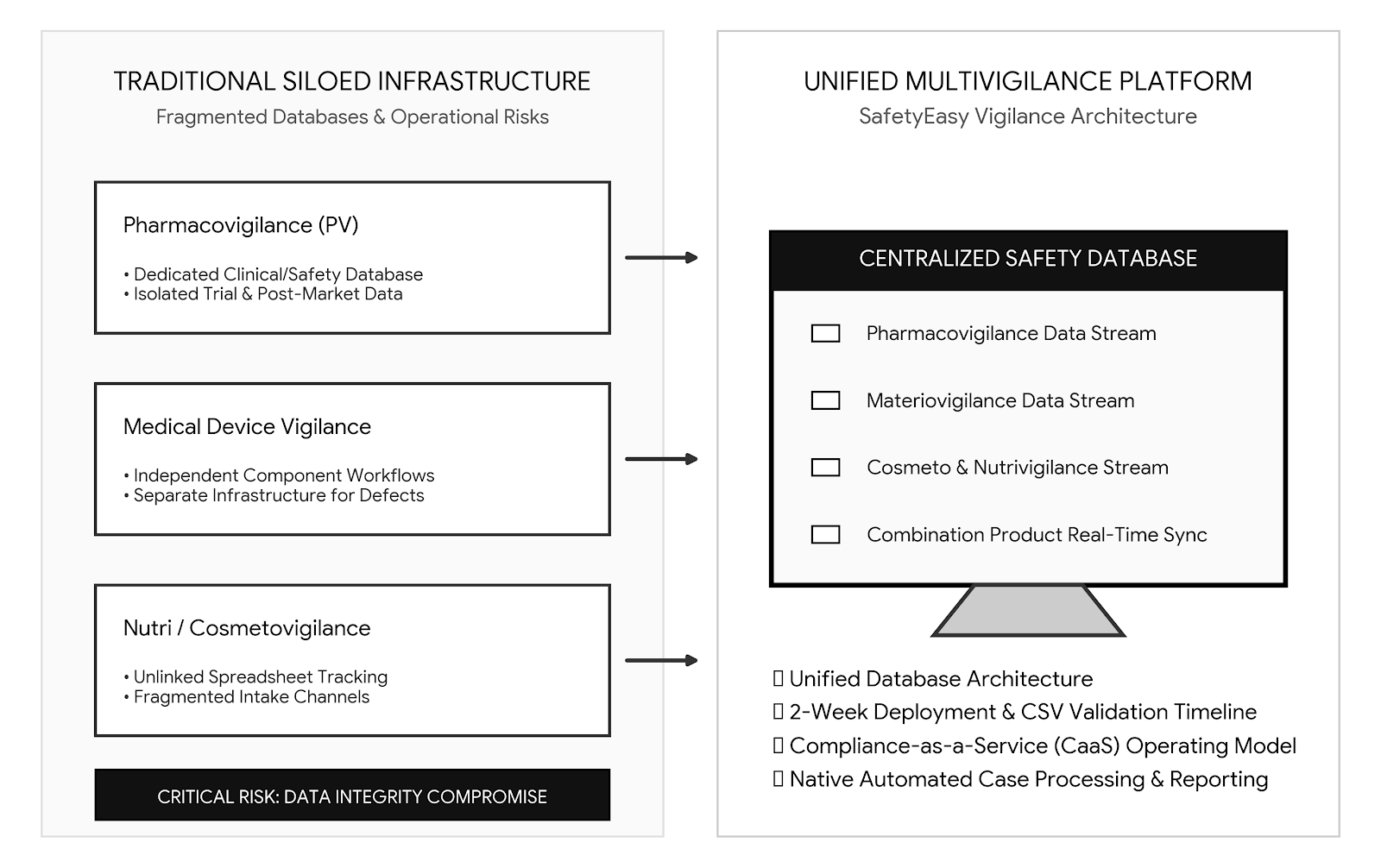
The Transition from Pharmacovigilance to Multivigilance in Modern Life Sciences
May 26, 2026
Managing safety data within the life sciences industry has historically followed a predictable framework. The drug safety department operates within a specialised database. The clinical team tracks trial irregularities in a separate infrastructure. C...
Learn more
Annex 11 Is About to Get Four Times Longer: What the EU's 2026 Rewrite Means for APAC Pharma
May 20, 2026
The European Commission opened consultation on the revised EU GMP Annex 11 back in July 2025. It closed on 7 October. The final text lands mid-2026. If this is the first you're hearing of it, you're not alone, and that's part of the problem.Most qual...
Learn more
CSA Is Not Optional Anymore: What FDA's Computer Software Assurance Guidance Means for Pharma in 202
May 15, 2026
When the FDA published its final Computer Software Assurance guidance in September 2022, most quality teams in APAC filed it under "interesting, deal with it later." Three years later has arrived. CSA is now the assumption baked into every fresh FDA ...
Learn more
APAC Pharmacovigilance in 2026: Why Speed and Compliance Are Finally on the Same Team
May 12, 2026
Three years ago, a clinical-stage biotech in Singapore asked us how quickly they could stand up a compliant safety database. The honest answer at the time was 'four to six months. Six months of meetings. Mountains of validation paperwork. Customisin...
Learn more
How to Prepare for an HSA Pharmacovigilance Inspection: A Step-by-Step Guide for Singapore Companies
May 04, 2026
If youve just received notice of an HSA pharmacovigilance inspection, the first thing to know is this: youre not in trouble yet, but how you spend the next few weeks will decide whether you stay out of it.The Health Sciences Authoritys Vigilance a...
Learn more
21 CFR Part 11 Compliance Checklist for APAC Pharma Companies: A Practical 2026 Guide
Apr 22, 2026
If you have ever sat across the table from an FDA inspector, or even just imagined doing so, you already know that 21 CFR Part 11 is the regulation that quietly decides whether your electronic systems pass or fail.And here in APAC, that conversation ...
Learn more
Automated Pharma Compliance in APAC: Why Valina CaaS & SafetyEasy® are the Ultimate Burden Removers
Feb 23, 2026
Let's be honest, in the life sciences and pharma world, "safety" is often just a polite word for "a mountain of paperwork."You buy a new software system, thinking it'll save you time, only to realize you now need three new employees just to manage th...
Learn more
Is Your ISMS Ready for 2026? Why ISO 27001 2022 is the New Minimum for Business Trust
Jan 14, 2026
In a digital landscape that changes by the hour, "standard" security is no longer enough. We are living in a world of rapid AI integration and sophisticated cyber threats where data integrity is the only thing standing between a company and a catastr...
Learn more
Why a Quality Management System Is a Must for Regulated Businesses
Jul 16, 2025
In areas where rules are strict, records need to be accurate, and inspections are important, having a Quality Management System (QMS) is not just a checkmark; it's essential for keeping everything running smoothly and reliably. Unfortunately, some bu...
Learn more
What's Next for Computerized System Validation: Top Trends for 2025 and Beyond
Jan 28, 2025
In the fast-changing world of life sciences, where rules are strict and technology moves quickly, Computerized System Validation (CSV) is evolving to keep up. For companies in pharmaceuticals, biotech, or medical devices, staying on top of these ...
Learn more
Regulatory Shifts Reshaping Life Sciences in APAC for 2025
Apr 08, 2025
The Asia-Pacific (APAC) region is witnessing a significant transformation in its regulatory environment, particularly across the life sciences industry. Governments and health authorities are actively refining their frameworks to encourage innovation...
Learn more
Mastering Computer System Validation Rules Across Borders
Feb 15, 2025
In the tightly regulated world of pharmaceuticals, biotech, and medical devices, ensuring your computer systems meet global standards is a must for keeping operations safe and compliant. At Valina Services, we know how tricky it can be to navigate th...
Learn more
Smarter Validation with Cerberus Testing: A Game-Changer for Regulated Industries
May 26, 2025
In industries like pharmaceuticals, biotech, and medical technology, where strict regulations govern every process, validating computer systems is a must. These systems, critical for GxP processes, need to ensure patient safety, product quality, and ...
Learn more
Why Validating Your Quality Management System Is Key for FDA Compliance
Feb 25, 2025
In the life sciences world, following strict rules is a must. Companies need to meet tough FDA standards to keep products safe, data accurate, and processes running smoothly. A key part of this is making sure your Quality Management System (QMS) ...
Learn more
SafetyEasy: Simplifying Drug Safety with Smart, Rule-Ready Solutions
Jun 09, 2025
In the rapidly expanding life sciences sector across Asia-Pacific, handling drug safety has become both crucial and challenging. Teams responsible for pharmacovigilance are dealing with stricter rules, varied local demands, and rising numbers of case...
Learn more